 The advent and improvement of NGS technology has revolutionized the scope to investigate repertoires ( 9). The highest throughput technology in genomics is currently next generation sequencing (NGS). Such region is called complementary determining region 3 (CDR3). The region translated from the junction determines antigen specificity. 
Additionally, somatic hyper-mutation (SHM) and insertion and deletion (INDEL) of nucleotides at V-D-J junctions can tremendously increase the junctional diversity ( 3, 8). Heterogeneous clonality in BCR repertoire is derived from the fact that recombination of V, D, J gene segment occurs at DNA levels independently in every B cell ( 8). Major challenge in BCR repertoire analysis arises from difficulties in interrogating astronomical diversity. Furthermore, the repertoire of in vitro phage antibody library can be explored by deep sequencing to accelerate antibody discovery without conventional screening ( 7). Examination of in vivo repertoire provides candidates of antigen-pecific monoclonal antibodies that can help us discover antibody drugs ( 5, 6). Thus, monoclonal antibody market has been rapidly enlarged ( 4). In clinical field, immunotherapy is leading a new paradigm in oncology. The repertoire analysis has expanded our horizons of understanding unseen diversity and complexity of the adaptive immune system. In immunology, emergence of powerful high-throughput methods such as parallel sequencing and mass spectrometry has allowed us to deeply investigate BCR repertoires. In recent years, research on BCR repertoire is not only an intriguing area in natural sciences, but also in pharmaceutical industries with significant impact. In this review, we will focus on BCR repertoire and interchangeably use both terms, BCR and antibody ( 3). In particular, B lymphocytes play pivotal role in humoral immunity through secreting BCR molecules, also known as antibodies and immunoglobulins, into body fluid. As a result, this immunological selection mechanism can lead to a highly personalized collection of lymphocytes in every individual which is called immune repertoire. Receptors that can recognize these antigens with high binding affinity are selected out of a huge receptor pool prepared by random mutations. Undoubtedly, antigens are extremely diverse spanning from infectious particles to self-peptides. Acquired immune responses can be initialized and achieved through highly specific recognition of antigen with cell surface receptors including B cell receptor (BCR) and T cell receptor (TCR) ( 2). Finally, we discuss limitations of current BCR-seq technologies and future perspectives on advances in repertoire sequencing.Īdaptive immunity is a specialized protective immune system in vertebrates against a variety of pathogens by utilizing B and T lymphocytes ( 1). Furthermore, we provide a summary of statistical frameworks and bioinformatics tools for clonal evolution and diversity. Due to the nature of massive sequence variation in BCR, caution is warranted when interpreting repertoire diversity from error-prone sequencing data. 
We also highlight preprocessing steps to remove sequencing and PCR errors with unique molecular index and bioinformatics techniques. These experiments should be carefully designed according to BCR regions to be interrogated, such as heavy chain full length, complementarity determining regions, and isotypes. 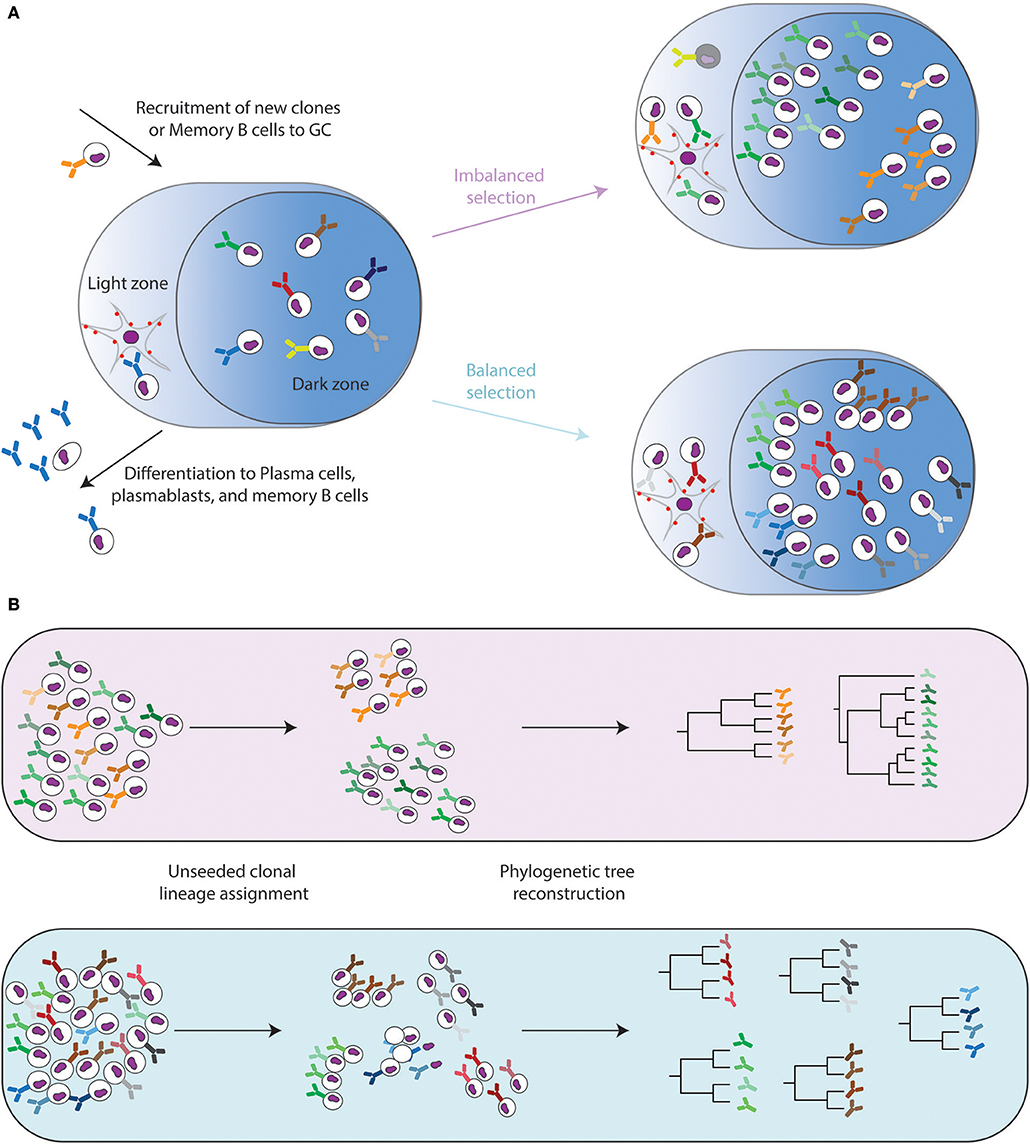
Here, we focus on B cell receptor (BCR) repertoire and review approaches to B cell isolation and sequencing library construction. Over the past several years, fast growing technologies for high throughput sequencing have facilitated rapid advancement of repertoire research, enabling us to observe the diversity of repertoire at an unprecedented level. As the repertoire shapes and represents immunological conditions, identification of clones and characterization of diversity are critical for understanding how to protect ourselves against various illness such as infectious diseases and cancers. Immune repertoire is a collection of enormously diverse adaptive immune cells within an individual. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
